Project Overview
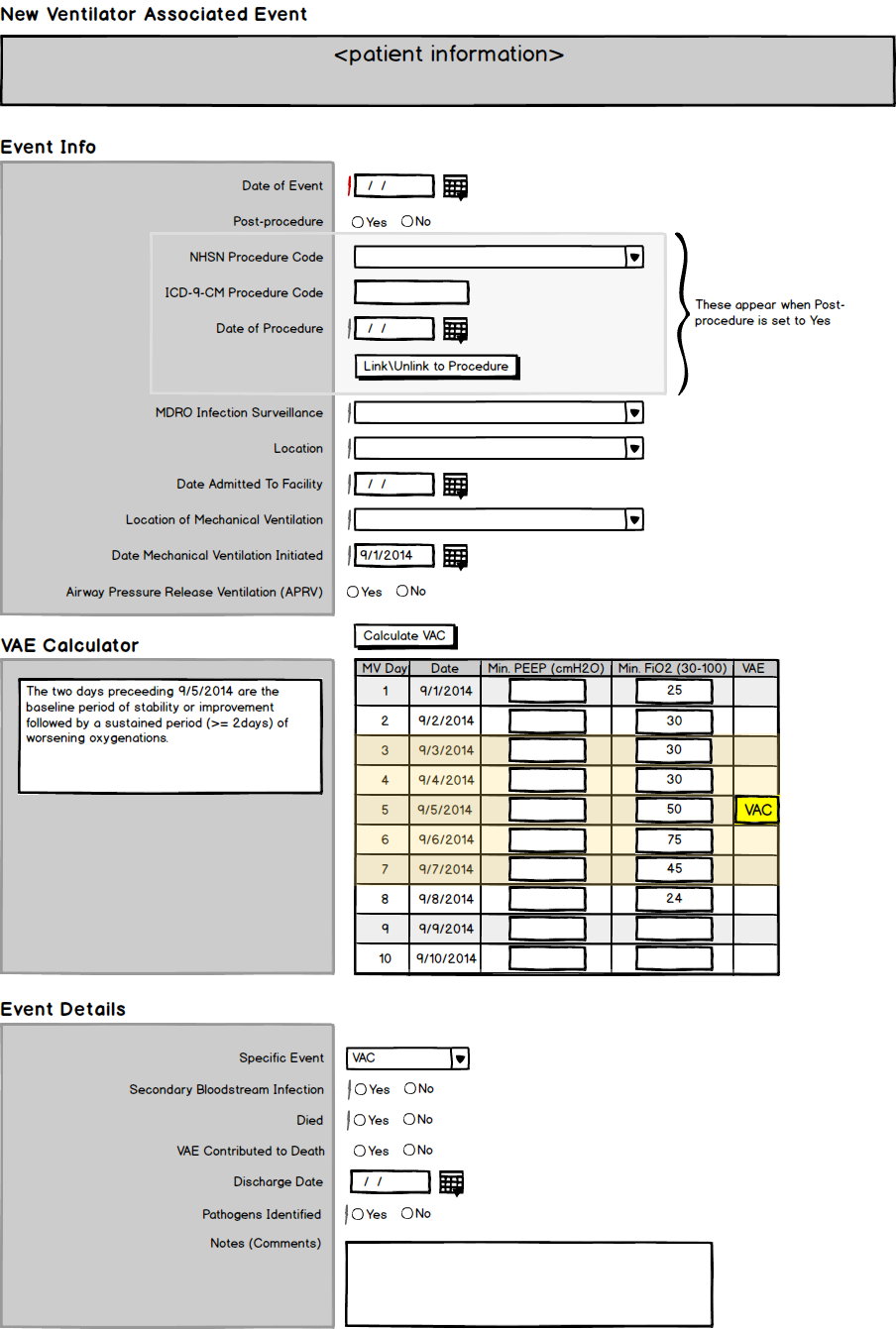
Pharmacy OneSource is owned by Wolters Kluwer and provides numerous SaaS products that empower clinicians to improve outcomes at the point of care. Along with real-time surveillance of all the hospital's systems, the Sentri7 product produces reports and documents for the Center for Disease Control's (CDC) National Healthcare Safety Network (NHSN) reporting of pathogens. What a hospital wants to do is show that a pathogen was not acquired at the facility (Hospital Acquired Infection or HAI), but was already present at admission (Community Acquired Infection or CAI.) This can be very tricky when patients are put on ventilators and the CDC has a very specific protocol that specifies what qualifies as an HAI or CAI and how this is to be reported. The objective was to take the current protocol and create a form that could be digitally sent to the CDC for NHSN reporting. This form would contain logic that would determine the type of Ventilator Associated Event (VAE) and then create the correct form to be submitted.
Challenges
The CDC will often update their requirements as often as every 6 months, so the forms need to be dynamic and capable of backward compatibility for up to a year. The requirements for specifying a VAE are so convoluted that the CDC has actually created an online application that helps nurses and respiratory therapists figure out what sort of report they need to fill out.
Process
Become a VAE expert
I read all the documents that the CDC supplies around VAE reporting. I interviewed our subject matter experts at Pharmacy OneSource. I then conducted 8 client interviews to determine how they reported VAEs and to gather Voice of Customer (VoC). I also gathered data on the type of ventilators they were using and if data was being collected from them and at what frequency.
Map out the process
Once I had all my data collected, I worked it into flow charts and created diagrams that outlined the process of determining the type of report we would need and the data required to fill that report. Further analysis revealed that a very short path could be made that quickly specified the type of report. I then took this to the SMEs and clients to vet its validity.